 Use your knowledge and skills to help others succeed.ĭon't be wasteful protect our environment. Please include it as a link on your website or as a reference in your report, document, or thesis. (Notice: The School for Champions may earn commissions from book purchases) Modern atomic theory goes beyond Dalton's views and includes concepts such as subatomic particles and isotopes. Although Dalton's theory remains valid, it also includes some misconceptions. The atomic theory of matter states that matter is composed of discrete units called atoms was created by chemist John Dalton in 1808. (See Structure of the Atom for more information.) Summary 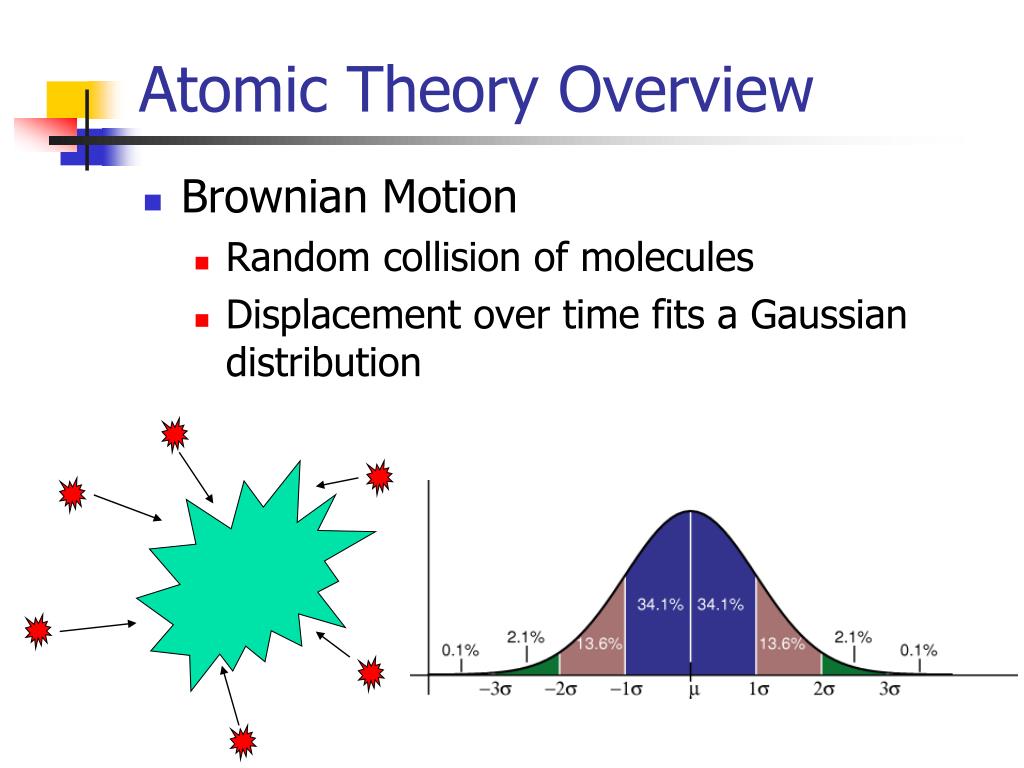
The model of the atom included the Bohr Model of a nucleus surrounded by electrons in orbit and Quantum Theory, where particles can exist as waves. IsotopesĪn element can have more or less neutrons in its nucleus. The atom was found to consist of a nucleus and subatomic particles. Brownian Motionīrownian motion showed atoms and molecules in motion. However, there have since been modern theories concerning the nature of matter and atoms. Modern ideasĭalton's atomic theory was a good start. Rearrangement of atomsĪ chemical reaction is a rearrangement of atoms.Īctually, in chemical reactions, atoms can be combined, separated, or rearranged. Compounds formed by combination of atomsĬompounds are formed by a combination of two or more different kinds of atoms.īut also two or more atoms of the same element can form molecules, although they are not called compounds. However, there are variations of atoms, called isotopes. Atoms of element identicalĪll atoms of a given element are identical in mass and properties. Basic rulesīasic rules of the theory-along with corrections to those rules-include: Matter made of atomsĪll matter is made of atoms, which are indivisible and indestructible.Īlthough all matter is made up of atoms, it was later found that atoms can be divided and even destroyed. His findings were based on experiments and the laws of chemical combination. A convenient unit of length for measuring atomic sizes is the angstrom (Å), defined as 10 10 metre. 
Approximately 50 million atoms of solid matter lined up in a row would measure 1 cm (0.4 inch). John Dalton, a British school teacher, published his theory about atoms in 1808. All atoms are roughly the same size, whether they have 3 or 90 electrons. 
It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
